- +49 201 43 70 97 0
- info@pakumed.de
- Mon - Fri: 8:30 - 17:00
TITAN PORT SPECIAL
TITAN-PORT DR
DRAINAGE
The TITAN-PORT DR allows repeated access to the pleural space
- The use of the corresponding special cannulas is obligatory.
- hypoallergenic
- High biocompatibility
- Particularly light (< 15 g)
- No chemical reactivity with injection solutions
- MR-conditionally safe up to 3 Tesla
- Secure twist and screw connection between chamber and catheter
- Suture holes for fixation to muscle fascia
- Compatible with non-coring port needles
- up to 3,000 punctures
- High pressure stability
- Self-closing special silicone
- Holds the port needle reliably in position
- Polyurethane or silicone: very flexible, but kink-resistant
- outstanding biocompatibility
- Suitable for long-term use
- free from latex, PVC and DEHP
- Length scaling for precise and safe implantation
- radiopaque
- Additional side hole for easier blood collection
- Conical tip for better insertion into the vein
- Catheter and port chamber are de-connectable
Description
Indication
Technical Data
Special Port Needles
Instruction for Use
Youtube Channel
Customer Feedback
Description

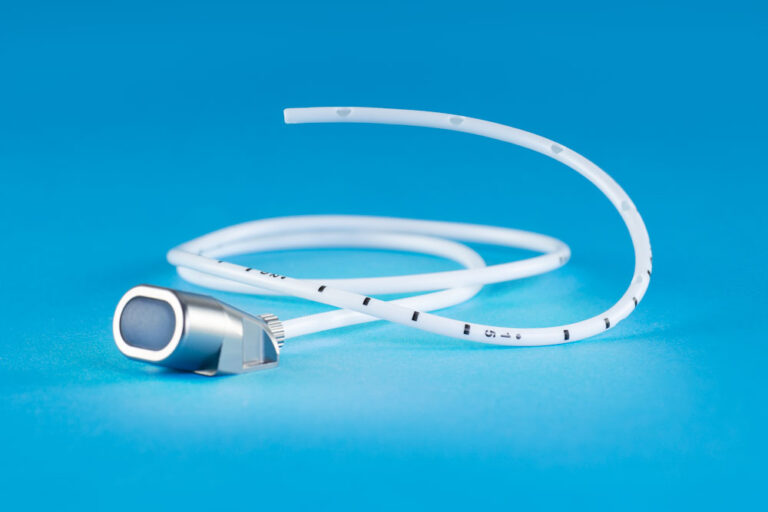
The TITAN-PORT DR (drainage) is a fully implantable port catheter system providing an access facility to the pleura cavity. The set contains a titanium port chamber with a self-sealing silicone membrane and a single-lumen polyurethane catheter for connection to the port chamber and fixing to the outlet tube with a screw. Each system also includes a port cannulae SFN 0930 G, a special puncture needle SFN 1835 B, an introducer set (consisting of an 18 G introducer, an 11 F introducer peel-away and a 0.89 mm guidewire), a tunnelizer, a vein lifter, a rinsing needle, user instructions and an implant card.
The polyurethane catheter (10 French, with an outer diameter 3.4 mm, an inner diameter 2.0 mm, total length 72 cm and internal volume of 0.31 ml per 10 cm) has at the distal end a fish mouth tip and ten side holes. If necessary the catheter can be shortened at the proximal (non-perforated) end.
TITAN port catheter systems are MR-conditionally safe. We will be happy to provide you with further information upon request.

Indication
The TITAN-PORT DR allows repeated access to the pleura cavity if necessary especially for drainage of blood, secretions and gases within the following indications:
- Pleural effusion
- Pleural empyema
- Chylo-, haemato- or pneumothorax
- Abcess
- Installation of therapeutics e.g. antibiotics, cytostatics or rinsing fluid
The advantage is a low risk of infection, simplified access and considerably improved quality of life for the patient as provided by a closed system. In particular, this significantly improves the patient’s quality of life and minimises the infection rate during access with pleural catheters.
Patient group: Patients with the need for long-term access to the pleura cavity.
Operators: Medical professionals (doctors, nurses) with appropriate qualifications.
Intended purpose:
The port catheter system is only used to pass liquids and medication into or out of the patient’s thoracic cavity. The product itself thus fulfils a physical property and has no medical, therapeutic effect.
Technical Data
| Art.No. | Port | Catheter | PZN | |||||||||||
| Material | Wide | Length | Height | Septum | Weight | Volume | Material | AD | ID | French | Volume | |||
| 111262 DR* Drainage | Titan | 20,6 mm | 25,5 mm | 16,7 mm | 118 mm2 | 15 g | 1,4 ml | Polyurethane | 3,35 mm | 2,0 mm | 10 with fish mouth tip, 10 side holes | 72 cm: 2,3 ml | 13837550 | |
*including introducer and tunnelizer
Special Port Needles
The TITAN-PORT DR allows repeated access to the pleural space when necessary, especially for drainage of blood, secretions or gases in the following applications, for example:
- Pleural effusions
- Pleural emphysema
- Chylo-, haemato- or pneumothorax
- Abzsessen
- Installation of therapeutics e.g. antibiotics, cytostatics or rinsing fluids
The advantage lies in the use of a closed system with longer retention time and lower infection rate. In particular, this significantly improves the patient’s quality of life and minimises the infection rate during access with pleural catheters.
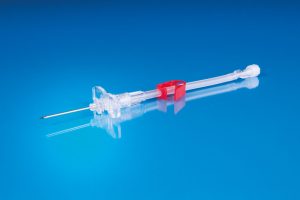

Instruction for Use
Youtube Channel
Here you can find all application videos for our products.
Customer Feedback