- +49 201 43 70 97 0
- info@pakumed.de
- Mon - Fri: 8:30 - 17:00
TITAN PORT SPECIAL
TITAN-PORT D
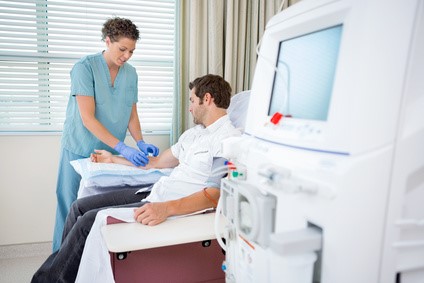
DIALYSIS
The TITAN-PORT D enables repeated alternative vascular access, e.g. for veno-venous dialysis in dialysis-dependent renal insufficiency.
- The use of the corresponding special cannulas is obligatory.
- Hypoallergenic
- High biocompatibility
- Particularly light (<22 g)
- No chemical reactivity with injection solutions
- MR-conditionally safe up to 3 Tesla
- Secure twist and screw connection between chamber and catheter
- Suture holes for fixation to muscle fascia
- Compatible with non-coring port needles
- High pressure stability
- Self-closing special silicone
- Holds the port needle reliably in position
- SFN Special port needle15 G – 1,000 punctures; 14 G – 500 punctures
- Polyurethane or silicone: very flexible, but kink-resistant
- Single or double lumen catheter
- outstanding biocompatibility
- Suitable for long-term use
- free from latex, PVC and DEHP
- Length scaling for precise and safe implantation
- radiopaque
- Catheter and port chamber are de-connectable
Description
Indication
Technical Data
SFN® Port Needles
Instruction for Use
Youtube Channel
Customer Feedback
Description

TITAN-PORT D (dialysis) is a fully implantable venous port catheter system as an access option for performing veno-venous dialysis.
There are 2 different systems available in complete sets:
the port catheter system with double-lumen PU catheter 14 F and the port catheter system with single-lumen silicone catheter 12 F:
- Double lumen dialysis port catheter system
The set includes two identical titanium port chambers with a self-sealing silicone membrane and two screws to fix the catheters to the outflow tubes, and a polyurethane catheter with an inflow and outflow connection to a port chamber. Each system is also supplied with two special puncture cannulas SFN 1835 B, a puncture cannula SFN 0930 G, an introducer set (consisting of an 18 G introducer cannula, a 15 F introducer peel-away and a 0.89 mm guide wire), a tunnelizer, this instructions for use and an implant card.
- Single lumen dialysis port catheter system
The set includes a titanium port chamber with a self-sealing silicone membrane and a screw to fix the catheter to the outflow tube and a single lumen silicone catheter with an inflow and outflow connection to the port chamber. Each system is also supplied with a special puncture cannula SFN 1835 B, a puncture cannula SFN 0930 G, an introducer set (consisting of an 18 G introducer cannula, a 13 F introducer peel-away and a 0.89 mm guide wire), a tunnelizer, these instructions for use and an implant card.
TITAN port catheter systems are MR-conditionally safe. We will be happy to provide you with further information upon request.

Indication
The TITAN-PORT D allows repeated access to the central venous blood vessel system for the following applications, for example:
– veno-venous dialysis for renal insufficiency requiring dialysis
– primarily
– as an alternative in case of failure of other access routes for long-term dialysis e.g. Cimino shunt
The advantage is a low risk of infection, simplified access and considerably improved quality of life for the patient as provided by a closed system.
Patient group: Patients in need of long-term central venous access to the vascular system.
Operators: Medical professionals (doctors, nurses) with appropriate qualifications.
Intended purpose:
The port catheter system is only used to pass blood and medication into or out of the patient’s vascular system. The product itself thus fulfils a physical property and has no medical, therapeutic effect.
Technical Data
| Art.No. | Port | Catheter | PZN | |||||||||
| Material | Wide | Length | Height | Septum Ø | Weight | Volume | Material | French | Volume | |||
| 111252 D-PU* Haemodialysis | Titan | 24 mm | 36 mm | 19,9 mm | 11,5 mm | 22 g | 0,8 ml | Polyurethane double lumen | 14 | longer leg: 2.3 ml shorter leg: 2.1 ml or 1.9 ml depending on the shortening | 01674462 | |
| 111255 D* Haemodialysis | Titan | 24 mm | 36 mm | 19,9 mm | 11,5 mm | 22 g | 0,8 ml | Silicone single lumen | 12 | Catheter 60cm: 0.49 ml/10 cm | 01674166 | |
*including introducer and tunnelizer
SFN® Port Needles
Only special non-coring port needles should be used to puncture the port membrane.
In order to achieve the blood volume necessary for dialysis purposes, correspondingly large-volume special port needles are required, e.g. SFN 1835 B or DPK 2035.
The needles exhibit a unique bevel and angle at the tip. This prevents punch defects at the membrane when the needle is inserted. Two suitable cannulas are included with each system. Appropriate accessories can be ordered separately if required.
| Art.-No. | needles | PZN | |||||||
| Ø mm | length mm | Gauge | Volume ml | Flow | Puncture frequency | unit | Extra | ||
| SFN 1835 B | 1,8 | 35 | 15 | 0,5 | with haemodialysis ~ 300 ml/min at gravity 225 ml/min | ~ 1000 | 25 pcs. | straight with Luer/Lock attachment With hose | 12363630 |
| DPK 2035 | 2,0 | 35 | 14 | 0,3 | with haemodialysis ~ 500 ml/min at gravity 300 ml/min | ~ 500 | 25 pcs. | mit Mandrin + nadelfreiem Adapter | 02429581 |
The SFN® special port needles are available separately for further applications:
Instruction for Use

Here you can look up all the info and its application in our PDF file.
Youtube Channel
Here you can find all application videos for our products.
Customer Feedback